Metal ion reactivity series8/23/2023 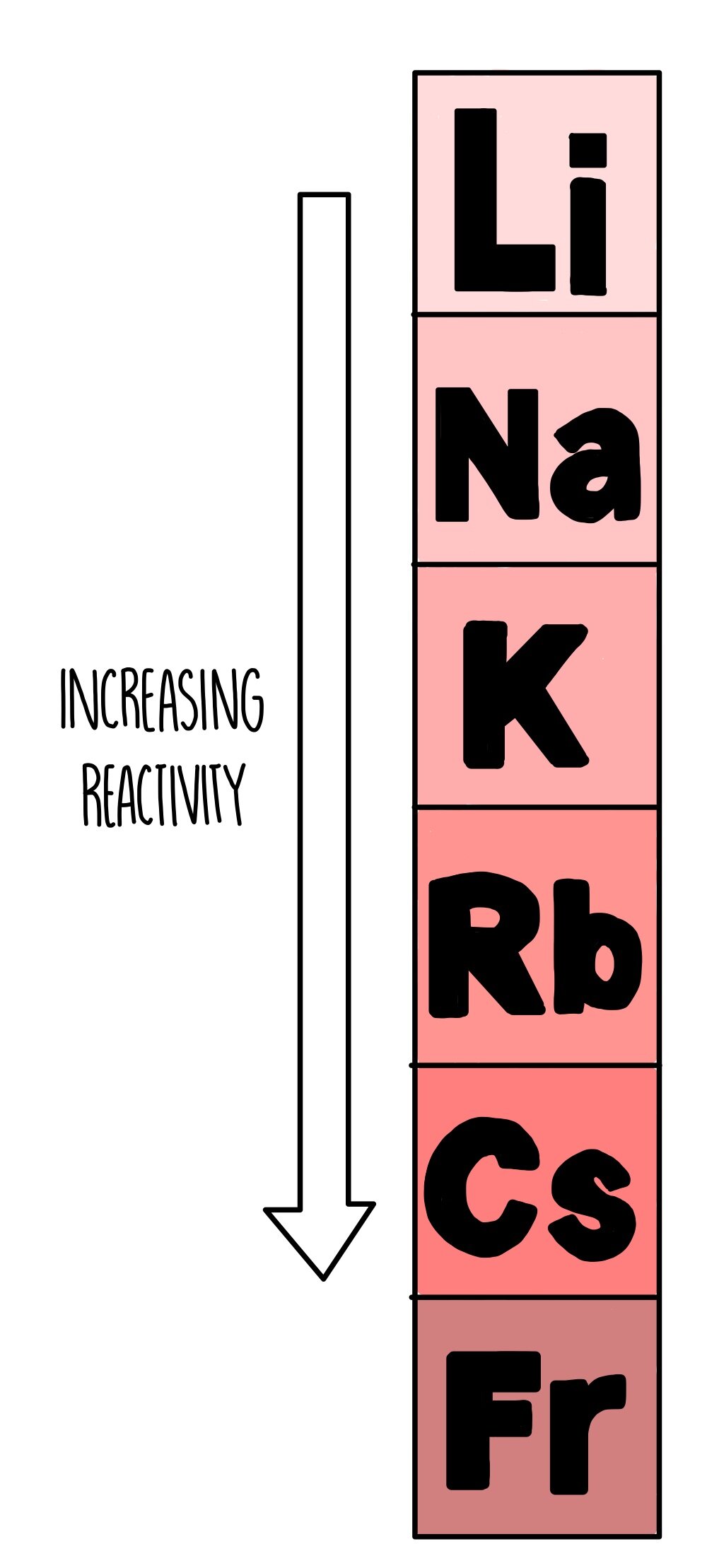
Group1 and group 2 elements are together called s-block elements. Alkaline earth metals are beryllium (Be), magnesium (Mg) calcium (Ca), strontium (Sr), barium (Ba) and radium (Ra). The group 2 elements in the periodic table are called alkaline earth metals. 
Alkali metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs) and francium (Fr). Alkali metals comprise group 1 in the periodic table along with hydrogen. The alkali metals are a series of chemical elements in the periodic table. Metals occupy the bulk of the periodic table. How are metals arranged in a periodic table? The more reactive a metal, the greater tendency it has to form a positive ion in a chemical reaction. 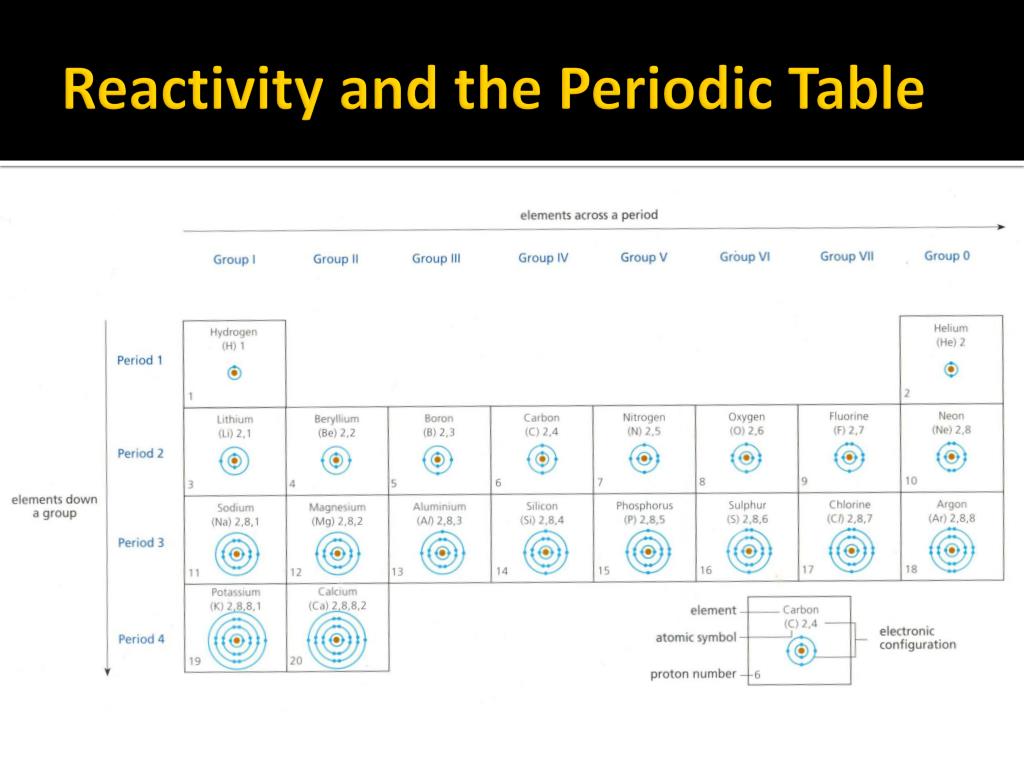 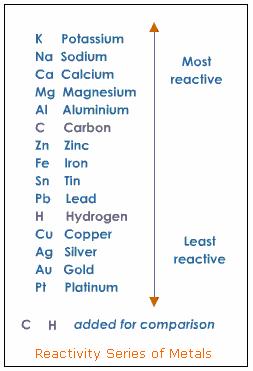
Most metals are electropositive in nature and the metal atoms lose electrons in chemical reactions to form cations. Metals are elements and are good conductors of heat and electricity. Predict what you will observe and the products formed if any.To observe the action of Zn, Fe, Cu and Al metals on the following salt solutions:Īrrange Zn, Fe, Cu and Al metals in the decreasing order of reactivity based on the above results.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
